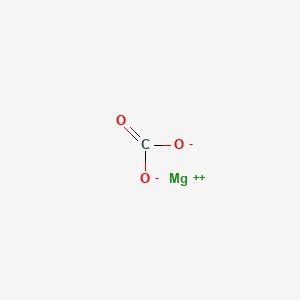
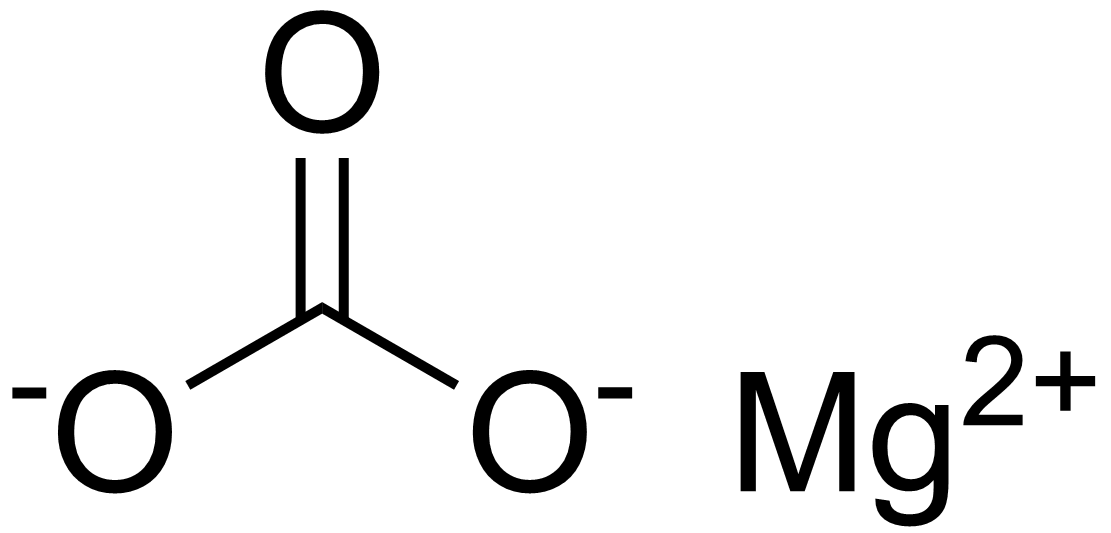
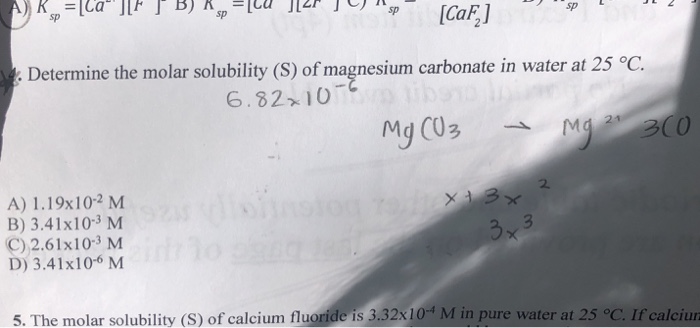Can you explain this sentence of my textbook: “It is because of high solubility product of mg(oh) 2 as compared to that of mg co3, that mg(OH) 2 is precipitated”? - Quora
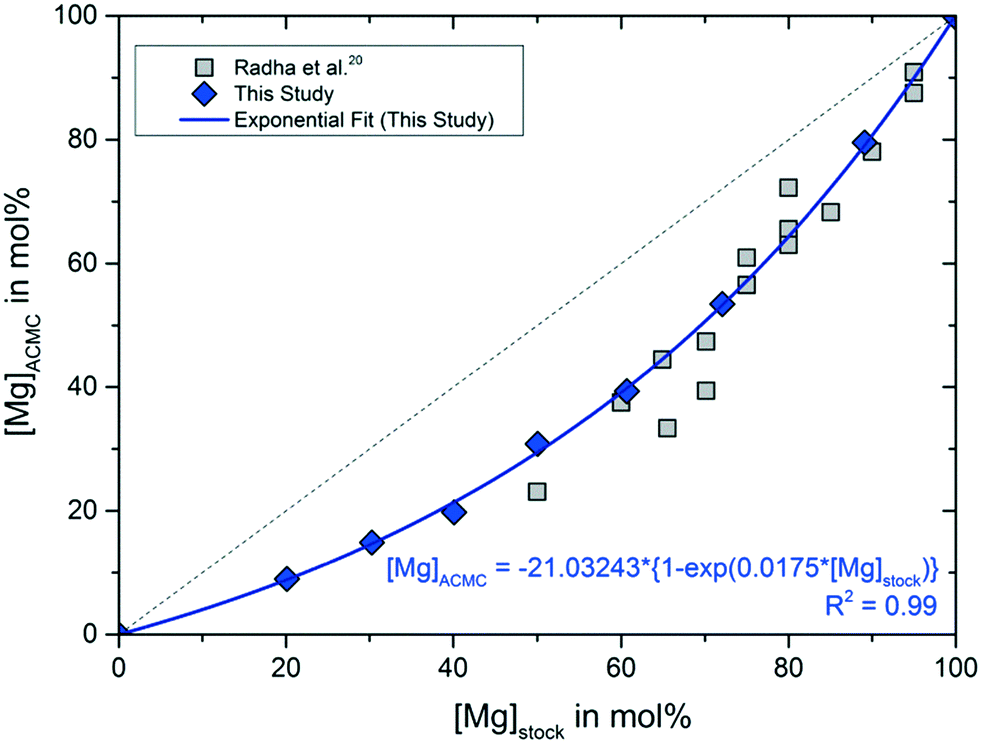
Solubility investigations in the amorphous calcium magnesium carbonate system - CrystEngComm (RSC Publishing) DOI:10.1039/C8CE01596A

SOLVED: Because these are similar, ie example apples to apples (from study guide) We can determine which one is least soluble. Which one has the lowest solubility in water? MgCO3 Ksp 6.8
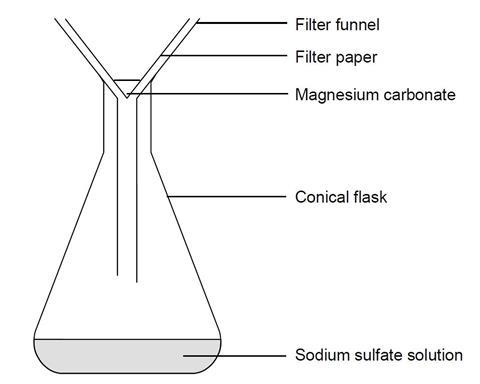
Making magnesium carbonate: the formation of an insoluble salt in water | Experiment | RSC Education

inorganic chemistry - Are lithium bicarbonate and magnesium bicarbonate both soluble in water? - Chemistry Stack Exchange
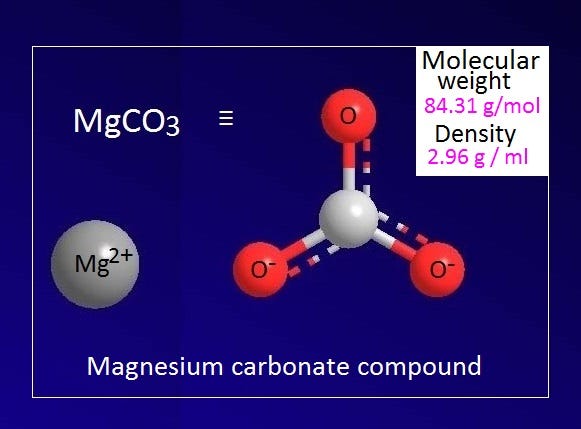
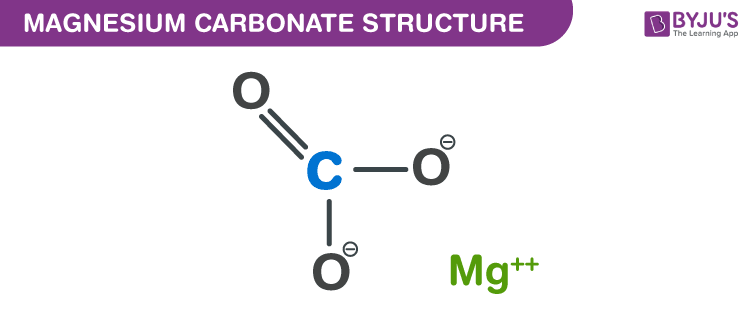
What is magnesium carbonate?. Magnesium carbonate is an inorganic… | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium