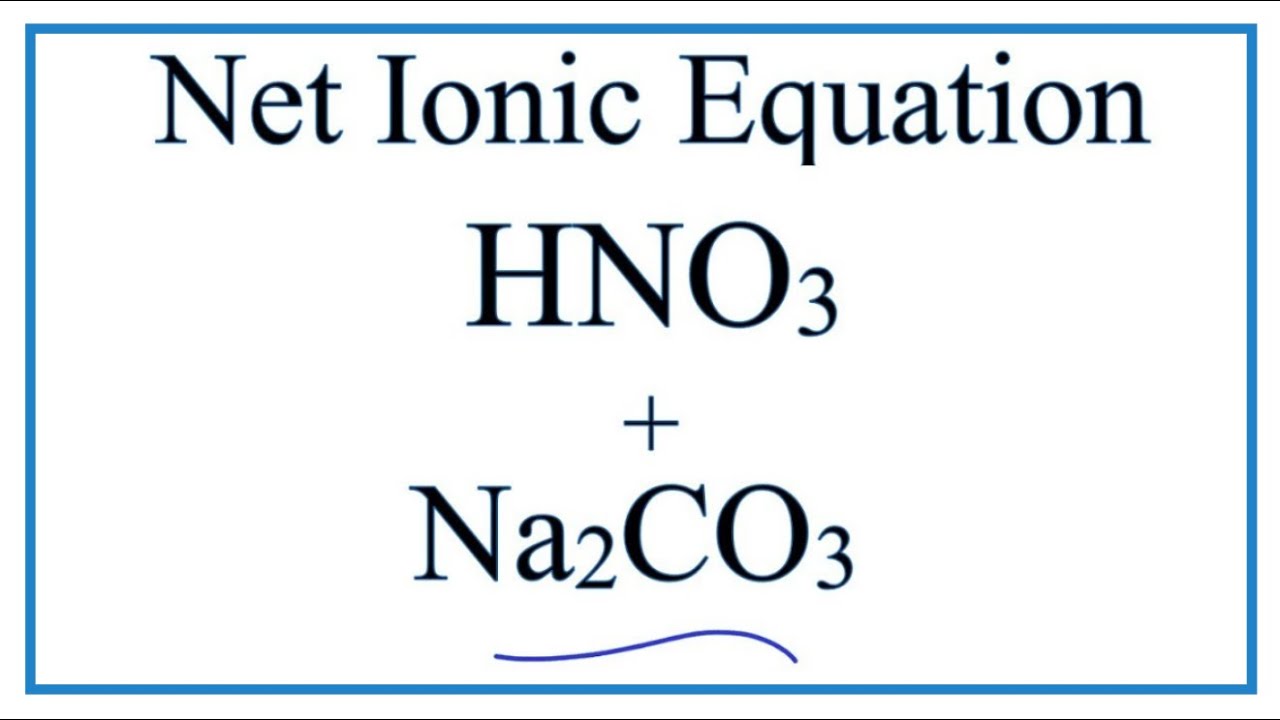SOLVED: In an accident, a solution containing 2.5 kg of nitric acid was spilled. Two kilograms of Na2CO3 was quickly spread on the area and CO2 was released by the reaction. Was

Effect of sodium carbonate, nitric acid, and acidic mixture on Akunsa... | Download Scientific Diagram

SOLVED: 1)Following a train accident, 2.0 x 104 gallons of 70.0% m/m nitric acid (d = 1.42 g/mL) was spilled; it was later neutralized using sodium carbonate. (a) Calculate the mass of
Write balanced chemical equation for the following : 1. Reaction of nitric acid with sodium bicarbonate. - Sarthaks eConnect | Largest Online Education Community
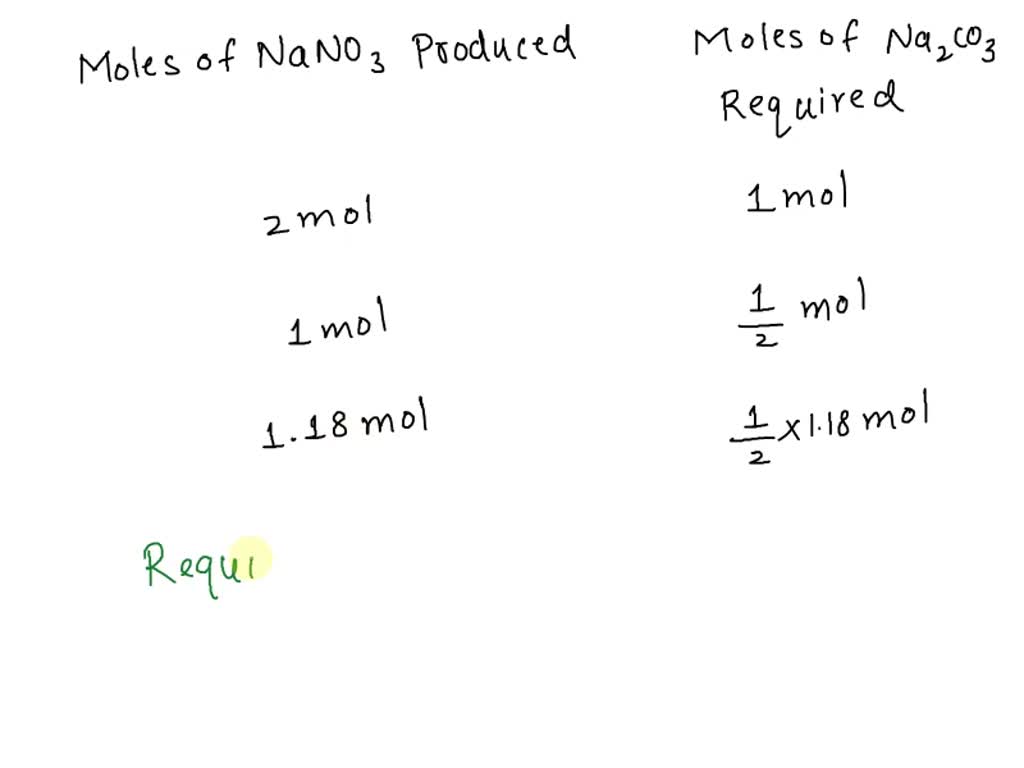
SOLVED: Sodium carbonate reacts with nitric acid according to the following equation: Na2CO3+2HNO3–>2NaNO3+CO2+H2O a. how many moles of Na2CO3 are required to produce 100.0 g of NaNO3? b. if 7.50g of Na2CO3

Sodium carbonate salt chemical structure, illustration - Stock Image - F027/9408 - Science Photo Library

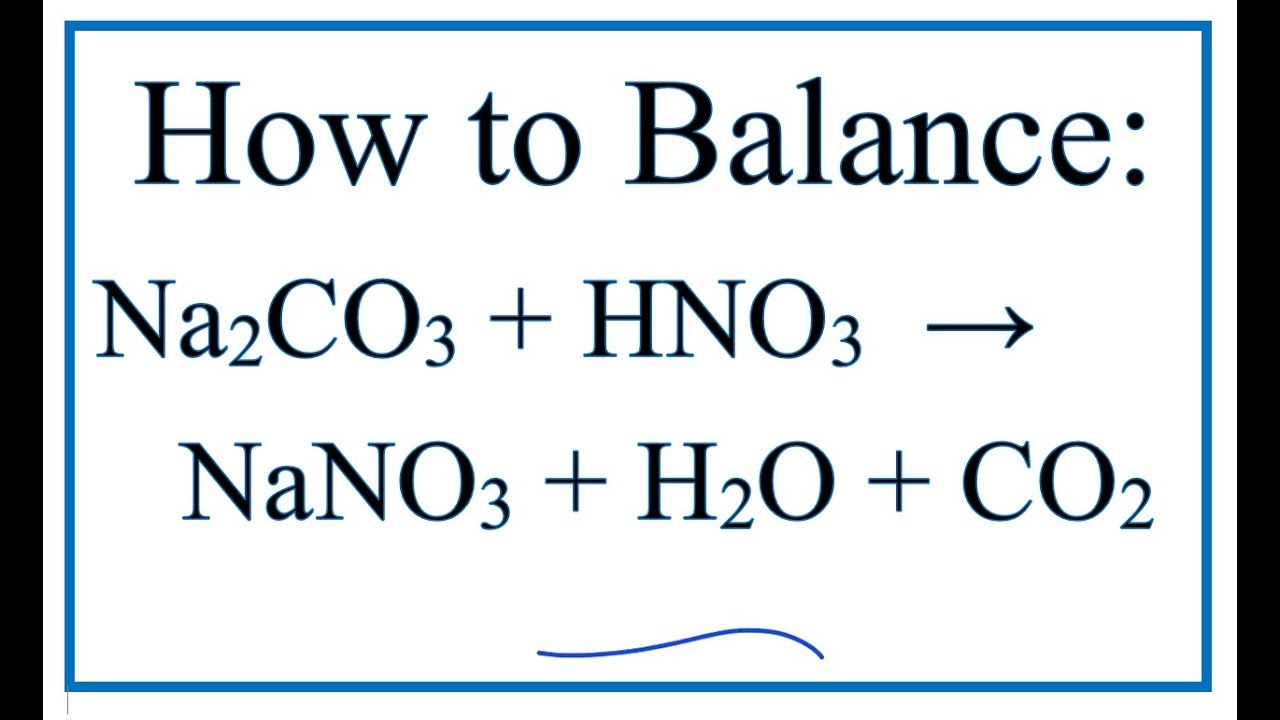





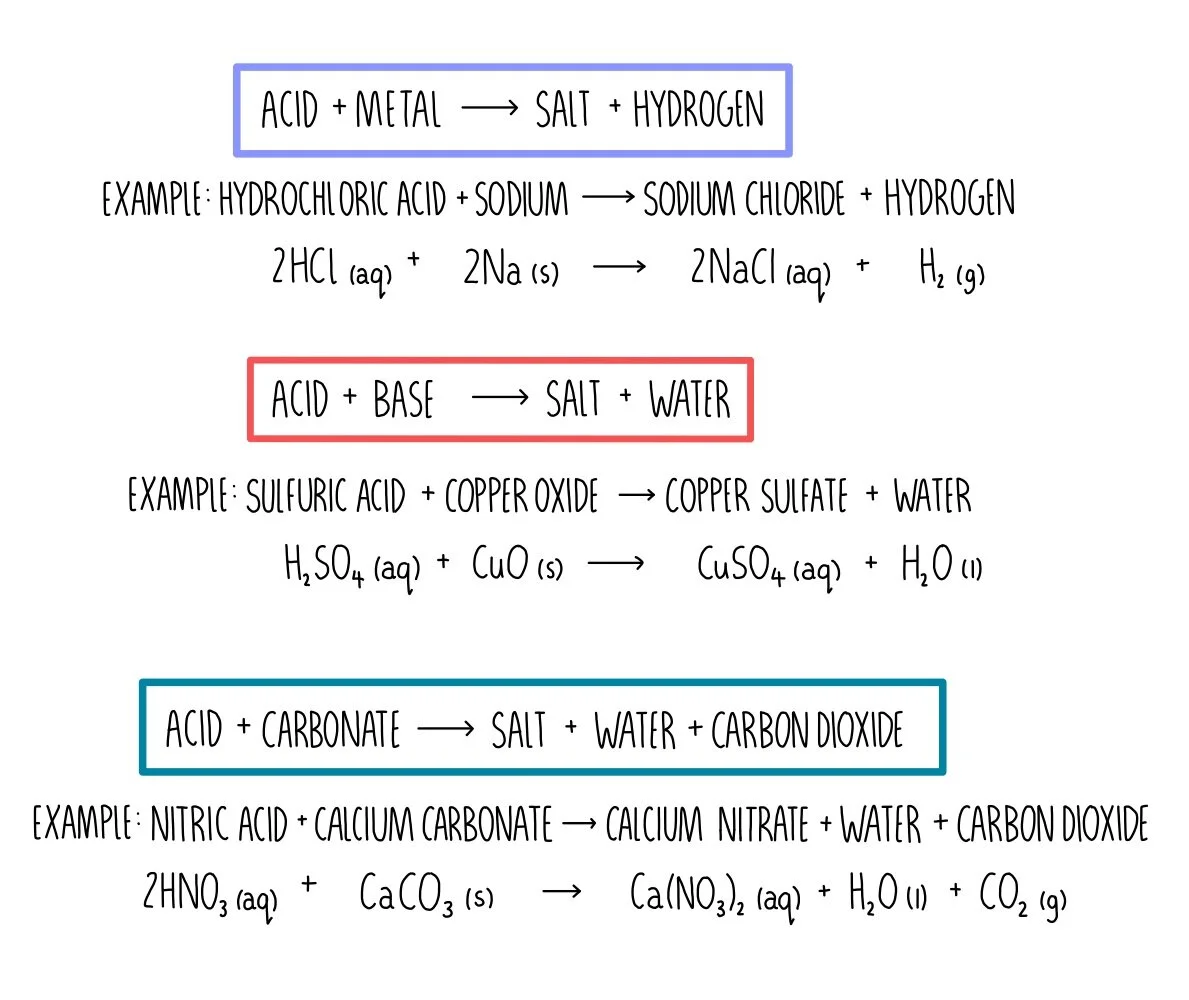

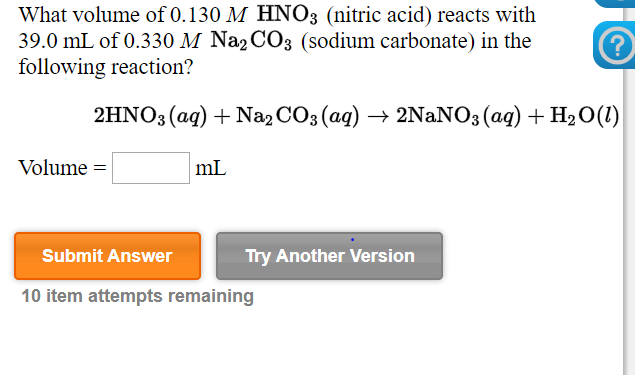

![ANSWERED] When nitric acid (HNO3) reacts with sodium... - Organic Chemistry ANSWERED] When nitric acid (HNO3) reacts with sodium... - Organic Chemistry](https://media.kunduz.com/media/sug-question/raw/59811332-1659634030.9246697.jpeg)